- Home
- About
-
Our Products
ELECTRIC HEATERS & HEATING ELEMENTS
- Tubular Heaters|Air Heaters
- Process Immersion Heaters|Tank Heaters
- Band Heaters
- Heat Tracers | Cable Heaters
- Silicon Rubber Heaters
- Heated Hose / Hose Pipe Heaters
- Etched Foil Heaters
View All
TEMPERATURE SENSORS & ACCESSORIES
- Thermocouples
- RTD Sensors
- Thermocouples Wires & Cables Accessories
- Temperature Transmitters
View All


- Technical
- Testimonial
- Contact us
Thermal Systems and Electric Heaters
Conduction is the transfer of heat energy through a solid material. Metals such as copper and aluminum are good conductors of heat energy. Glass, ceramics and plastics are relatively poor conductors of heat energy and are frequently used as thermal insulators. All gases are poor conductors of heat energy. A combination of expanded glass or ceramic fiber filled with air is excellent thermal insulation. Typical conduction heating applications include platen heating (cartridge heaters), tank heating (strip and ring heaters), pipe tracing and other applications where the heater is in direct contact with the material being heated.
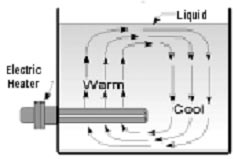
Convection is the transfer of heat energy by circulation and diffusion of the heated media. It is the most common method of heating fluids or gases and the most frequent application of electric tubular elements and assemblies. Fluid or gas in direct contact with a heat source is heated by conduction causing it to expand. The expanded material is less dense or lighter than its surroundings and tends to rise. As it rises, gravity replaces it with colder, denser material which is then heated, repeating the cycle. This circulation pattern distributes the heat energy throughout the media. Forced convectionuses the same principle except that pumps or fans move the liquid or gas instead of gravity.
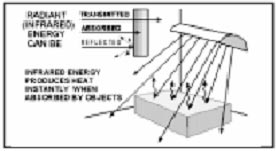
Radiation is the transfer of heat energy by electromagnetic (infrared) waves and is very different from conduction and convection. Conduction and convection take place when the material being heated is in direct contact with the heat source. In infrared heating, there is no direct contact with the heat source. Infrared energy travels in straight lines through space or vacuum (similar to light) and does not produce heat energy until absorbed. The converted heat energy is then transferred in the material by conduction or convection.
All objects above “absolute zero” temperature radiate infrared energy with warmer objects radiating more energy than cooler objects. Infrared energy radiating from a hot object (heating element) strikes the surface of a cooler object (work piece), is absorbed and converted to heat energy. Paint drying by radiant heaters is a typical application of infrared heating. The most important principle in infrared heating is that infrared energy radiates from the source in straight lines and does not become heat energy until absorbed by the work product.








